Request a quote.

The necessity of swimming pool water treatment
Various contaminants are introduced to the swimming pool by swimmers and the surrounding environment. The contaminants are a variety of microorganisms ranging from waterborne infectious viruses and bacteria, fungal contamination, to dissolved and undissolved matter (mostly organic). Poor maintenance can result in low disinfectant levels, allowing the spread of various germs and microbes that can cause severe diarrhea as well as skin and respiratory problems or other recreational water illnesses (RWIs).
To ensure a healthy swimming pool environment, an effective and continuous water treatment solution needs to be in place.
Mellifiq has extensive knowledge of water disinfection, and Ozonetech RENA Water Solutions offer the best applications for efficient and sustainable water treatment for swimming pools.
Other than enabling a healthy swimming pool environment, our goal is to offer a more efficient and sustainable solution that helps you reduce your energy costs, improve your business profitability and output quality, and to lower your swimming pool’s environmental impact.

Common challenges in swimming pool water treatment.
Low quality swimming water
Different methods have been used to reach a sufficiently high water quality. Among the most common methods are filter screens and sand filtration, chemical treatments (e.g. for flocculation and water softening), and disinfection with chlorine, UV and ozone. Different technologies are often combined depending on contaminant level and desired water quality. Mellifiq offers all required technologies for superb pool water quality, including:
- Saniray UV reactors with low and medium pressure lamps, complete with ballast and controls for any flow
- Turn-key ozone systems under our Ozonetech brand
- Water Maid FlexKarb multimedia filter systems
- Chemical dosing systems
The by far most common disinfection method today is chlorination. However, chlorination is also associated with complications regarding health effects among swimmers. Contrary to popular belief, chlorine does not kill all germs instantly. Many germs are also tolerant to chlorine and once they get in the pool, it can take anywhere from minutes to days for chlorine to kill them. Swallowing just a little water that contains these germs can make swimmers really sick.

Health risks with water chlorination
In 2014 the Scientific Journal Environmental Science and Technology published an article which shows that uric acid, (and chemically similar compounds from body fluids) and chlorine reacts and form some potentially harmful chemicals. These harmful chemicals are known as nitrogen-containing disinfection byproducts (N-DBPs) such as cyanogen chloride (CNCl) and trichloramine (NCl3).

These two compounds, even at low levels in swimming pools, are associated with mucous, eye and throat irritation and respiratory damage which may prevent swimmers from using the swimming pool. At high levels, they can adversely affect the nervous and cardiovascular systems. Other known by-products of chlorination are trihalomethanes (THMs) that cause other severe health damage. By using other disinfectants than chlorination these effects can be mitigated.
Ozone treatment on the other hand is more sustainable and enables the lesser use of chlorine, reducing water consumption, energy demand, and mitigating negative health effects caused by chlorine byproducts. Since ozone is produced from ambient air, the raw material is free resulting in lower costs of purchasing, transportation, handling, storage, waste handling and the residue removal of the chlorine treatment.

Existing treatment solutions
A standard design for swimming pool water treatment is typically based on the following treatment steps; filtration, disinfection, chemical treatment, and water replacement.
Filtration is commonly performed in two stages where the first stage removes larger pollutants like hairs and the second stage removes small particles.
Disinfection leads to deactivation of microorganisms and chemical decomposition of organic material. This is commonly achieved by chlorination of the water treatment stream, but can also be achieved by ozonation and UV-treatment. These different alternatives all have their advantages and drawbacks.
Additionally, chemical treatment is often used to soften the water (i.e. remove calcium and magnesium ions) or for flocculation (i.e. neutralization of negatively charged particles) which causes them to form larger particles that can be removed by filtration.

Our solution.
Ozonation as a modern alternative
When using an Ozonetech system, ozone is generated in-situ unlike traditional chemicals used for disinfection and cleaning, such as chlorine. Ozonation utilizes naturally occurring oxygen, which eliminates the need for chemical handling procedures. This means that there is no purchasing, no transportation, no handling, no storage, no waste and no residues involved like there is when using chemicals. Ozone is produced by imposing a high voltage across a dielectric discharge gap (Corona Discharge) which ionizes oxygen atoms and forms ozone molecules. Ozone is a much stronger oxidant than chlorine and once applied, ozone instantly reacts with contaminants, leaving no byproducts other than oxygen.
The main mechanisms for ozone disinfection include the destruction of microorganism cell walls, radical oxidation reactions, decomposition of nucleic acids (DNA and RNA), and breakage of carbon-nitrogen bonds (which are essential in most organic compounds like e.g. proteins). Two main factors influence the effectiveness of ozone disinfection, namely the contact time and ozone concentration.
At Mellifiq we are specialists in ozone treatment and know just how to tackle your swimming pool challenges and to ensure a cost-efficient way to disinfect and purify your swimming water.

The treatment process in swimming pools
The feed-gas to the ozone generator is added either in the form of purified oxygen or atmospheric air. Purified oxygen can be delivered to the swimming pool site, but is preferably also produced on-site from atmospheric air with an air separation unit generating a 93% oxygen feed.
Using a high-purity oxygen feed in combination with an effective cooling system, we offer a highly compact ozone generation design. We supply Ozonetech systems with production capacities ranging from 5-5000 grams O3/hour.
Ozone gas is effectively dissolved into the swimming pool waterside stream using venturi injection. This way, as much ozone as possible is transferred into the water for subsequent disinfection in the reaction tank as depicted in the process flow diagram below.
The dissolved ozone is evenly distributed into the ozone reaction tank. This is where the disinfection process takes place. It is important to provide enough contact time to achieve efficient disinfection results. To optimize the disinfection process three key parameters have to be controlled, namely ozone concentration, contacting, and reaction time. The final step in the ozonation process involves ozone destruction of the off-gas to ensure a healthy working environment for operators.


Disinfectant overview and comparison
To give an overview of the most common treatment alternatives the benefits and challenges of ozone treatment, UV-treatment, and chlorination were compared in the table below:
Benefits and challenges of three typical disinfectants:
| Benefits | Challenges | |
|---|---|---|
| Chlorination |
|
|
| UV-treatment |
|
|
| Ozone treatment |
|
|
Disinfectant efficiency comparison
Based on CT-values for virus disinfection provided by the EPA, a graphical comparison is made between chlorine, chloramine, chlorine dioxide, and ozone. A low CT-value represents an efficient disinfectant.
CT-value comparison for main chemical disinfectants.

Because of the high CT-value of chloramine, an additional figure is presented below to show the relationship between only ozone, chlorine dioxide, and chlorine.
CT-value comparison for main chemical disinfectants (except chloramine).

The graphs clearly show the small amounts of ozone needed for disinfection, placing ozone on top as the most effective commercial disinfectant.
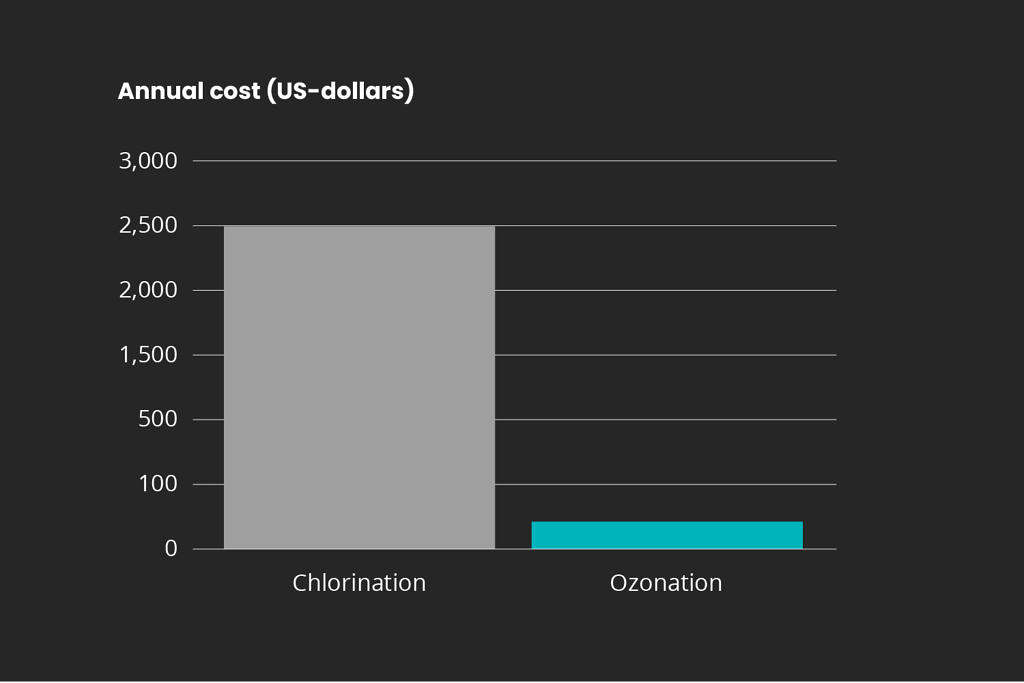
Disinfectant cost comparison
Estimated cost comparison between hypochlorite treatment and ozonation is shown below based on a 25 × 25 m commercial swimming pool which is operated 350 days per year. The comparison includes the hypochlorite chemical costs for chlorination and the power consumption costs for an ozone generator coupled with an oxygen concentrator.
Hypochlorite chemical cost vs. ozonation power cost
Our advantages
We offer premium state of the art technology with high reliability and efficiency, low energy consumption, and low maintenance cost. Other advantages include:
- Long lifetime of the ozone generator. This is partly due to the effective cooling system and the fact that concentrated, filtrated, and dried oxygen is fed to the generator.
- We offer turn-key facilities, ensuring reliable process operation.
- Compact design and the possibility to increase disinfection capacity due to modular generator design.
- High ozone concentration, maximizing treatment effectiveness.
Process design
The equations presented in the following paragraphs may be used to estimate the capacity requirements for a potential ozonation process for swimming pool water treatment.
Treatment capacity
To determine the capacity requirements of the water treatment system the most important factors include: swimming pool size, bathing load, pool type, and pool temperature.

To calculate the flow rate/capacity of the treatment system the following equation may be used. T is the time it takes for the equivalent of the entire swimming pool volume to be recirculated in the water treatment system. A value of d/T = 0.55 represents a “worst-case” value and this value will be used for further reference. Also, an f-value of 0.8 is used as it is a common value for Olympic pools.

Ozonation is applied to a bypass stream which is usually in the range of 25% of the main water treatment flow. This gives the following expression for the flow rate through the ozonation system, “Q(byflow)”, which requires ozonation.

For 28 ⁰C or 33-35 ⁰C the water requires an ozonation of 0.8 and 1.2 mg/l respectively. The contacting equipment allows for about 90 % dissolution efficiency of the generated ozone. However, a dissolution efficiency of 0.8 may be used for extra margin. Furthermore, the ozone generator capacity decreases over time. Hence, a decrease in capacity of 10 % can be used (to be conservative). To estimate the required ozone production, “Q(O3)”, of the generator the following formula could be used.
Reaction tank
To estimate the size of the reaction tank a minimum reaction time of 2.5 minutes should be used given an ozone concentration of 1 mg O3/L. This is equivalent to a CT-value of 2.5 (mg · min/L) which is definitely safe and sufficient for swimming pool water. As an example, a by-flow (to the ozonation system) of 60 m3/h requires a reaction tank volume of 2.5 m3.
Recommended products
Ozonetech RENA Vivo series
The Vivo segment offers the following operational enhancements:
- Eliminate all disinfection agents used for treatment or sanitation
- Reduce energy requirements by replacing chemicals with ozone sanitation
- Ensure completely microbial free water
- No handling of chemicals, transport and costs
- Treat complex wastewater substances on site
- Chemical-free feed and process water quality assurance

Recommended products
Water Maid
- Pressurized multimedia and adsorption systems
- Automatic backwashing
- Wide range capacity
- Highly efficient polishing for complete removal

Recommended products
Ozonetech UV water systems
- Complete UV reactors with ballast and control
- Single and multiple lamp configuration
- High quality flanged stainless steel reactor
- Low and medium pressure lamps
- Low maintenance

Recommended products
Ozonetech RENA Pro series
The Ozonetech RENA Pro segment offers the following operational enhancements:
- Highly efficient ozone generation reduces energy consumption and environmental footprint
- Stable and reliable ozone production over time
- High-quality feed-gas used (concentrated, dry oxygen from oxygen generator)
- Pure ozone output. No by-products such as NOx, no nitric acid (HNO3) nor salt formation; extending longevity of the system
- Efficient heat management (liquid cooling) for stable and reliable operation, also extending longevity of system
- Minimized maintenance due to clever design; pure, dry oxygen feed-gas and liquid cooling
- Teflon tubing for ozone delivery
- Rugged design for harsh environments

Related reference project.
More about chlorination, UV and ozonation
So called “chlorine”, in colloquial language, actually covers an entire group of substances. Common chlorine compounds are e.g. chlorine gas, sodium hypochlorite (liquid), calcium hypochlorite (granular), in situ electrolysis of NaCl solution, chlorinated isocyanurates (stabilized chlorine), and chlorine dioxide. These chlorine substances all share the property of forming free chlorine in water solution which is readily available for water disinfection. Free chlorines are typically hypochlorite ions (OCl-) and hypochlorous acid (HOCl).
In the chlorination process the level of chlorine has to be balanced to enable sufficient disinfection and at the same time minimize discomfort for swimming pool users. The main discomfort issues include mucous tissue irritation. According to the WHO the level of free chlorine should not exceed 3 mg/L in public and semi-public swimming pools. The maximum combined chlorine level for all temperatures at pH 7.2 – 7.6 should not exceed 0.4 mg/L. All chlorine based chemical treatments share the same problem; they all lead to buildup of combined chlorine which is the source for the above mentioned discomfort. Therefore, when using chlorination fresh makeup water has to be added regularly to the system to dilute and maintain an acceptable level of combined chlorine. When using chlorinated isocyanurates the maximum level of cyanuric acid proposed by the WHO is 100 mg/l.

UV-treatment
Ultra-Violet radiation is another technology used for swimming pool water treatment. A UV-lamp is used to generate radiation in the UV-spectrum which efficiently deactivates microorganisms, virus, and algae by physically destroying the DNA. It is often used as a complement to chlorination which then enables a reduction of chlorine consumption. On the other hand, UV-radiation also breaks down some of the chlorine which therefore has an increasing effect on the demand for chlorine.
Ozonation
Ozone is generated in-situ unlike traditional chemicals used for disinfection and cleaning. Ozonation utilizes naturally occurring oxygen which eliminates the need for chemical handling procedures. Ozone is produced by imposing a high voltage across a dielectric discharge gap (Corona Discharge) which ionizes oxygen atoms and forms ozone molecules. Ozone is a much stronger oxidant than chlorine and once applied ozone instantly reacts with contaminants, leaving no byproducts.
The main mechanisms for ozone disinfection include: destruction of microorganism cell walls, radical oxidation reactions, decomposition of nucleic acids (DNA and RNA), and breakage of carbon-nitrogen bonds (which are essential in most organic compounds like e.g. proteins). Two main factors influence the effectiveness of ozone disinfection, namely the contact time and ozone concentration.


